Thermapen ONE: The Ice, the Candy, the Oil, and the Racetrack
If you’re reading this, you probably already own a Thermapen® ONE. It goes without saying that we have…several. If you own one and have used it, you’re probably already hooked on the experience of unmatched accuracy and fast-as-thought readings, and endless versatility. But there is a question that some new Thermapen users have about Thermapen’s speed. When using Thermapen on hot oil or syrup for candy, for instance, they notice that the super-speed they’re used to is slightly less super. What’s going on? Isn’t this supposed to give one-second readings? No, your thermapen is not defective. Let’s get into it.

Get the Fastest Temperature Tool Available:
What it takes to take a temperature
To begin, we need to talk about how temperatures are taken. And this brings up a weird point. We never actually take the temperature of a thing; we actually take the temperature of the thermometer.
All of thermometry depends on the principle of equilibrium—that when two things are part of the same system, they come to the same temperature. We put a thermometer in a steak, the thermometer comes to thermal equilibrium with the steak, and we read the thermometer’s temperature at that equilibrium point. No matter what we’re temping, we must wait for the thermometer to equilibrate. In fact, you can see thermal equilibration more easily with a slower thermometer. On a dial thermometer, for instance, you can see the temperature rise or fall as the thermometer’s bimetallic strip heats or cools. (Equilibration often happens too fast to notice with Thermapen.)
So the speed at which we can read a temperature depends on how quickly the thing that we’re measuring can heat our thermometer. Now you may be wondering, how exactly does that work?

Phlogiston, even though it’s wrong
To build a bridge of understanding here, we can turn to an old and incorrect model of heat from the late 100’s called phlogiston theory. This theory supposed that heat was an invisible substance, which could flow from one object to another or escape into the air. When something felt hot, it had lots of phlogiston in it. When something was cold, it had less. A rise in temperature would correspond with phlogiston filling it up. When phlogiston flowed out, the object cooled.
We now know this theory is wrong, but it can be handy for helping us understand how heat works. In the past, they would say that it takes a certain amount of phlogiston entering our thermometer to raise the temperature, or a certain amount flowing out of it to decrease the reading. So, using that model as a basis for understanding, we can look at what really happens.

Specific heat capacity and conductivity: what’s really going on
First, “heat” isn’t stuff. It’s the ability of stuff to perform work. And Heat isn’t temperature, either. When something receives more heat, its temperature goes up, but different things get hotter at different rates for the same amount of heat.
Let me explain.
If you put your hand in an oven that is 350°F (177°C), it’s quite warm. You wouldn’t want to hang out there, but the air isn’t going to burn you very quickly. If you grab the steel pot handle that has been in that oven for 30 minutes, you’re going to yell and run for the burn spray. The air and the pot are at the same temperature, but the pot holds far more heat at that temperature.

Specific heat
Specific heat tells us how much thermal energy a given mass of substance can hold per degree of temperature. Water has a very high specific heat. Oil? Not so much.
One pound of water holds about 2.5 times as much energy as one pound of vegetable oil at the same temperature. A pot of oil, therefore, has less energy in it to cause the temperature of our thermometer probe to rise. It WILL come to temp, but the oil will have to give up a higher percentage of its energy to do it. And it will do it more slowly.1
Even water-like substances—the syrups made in creating homemade candies—have lower specific heat capacities. The sugar interferes with the water’s ability to hold and pass heat around, dulling the thermal effect of the temperature.

Thermal conductivity
So we come to the final thermal principle that explains thermometer speed. Materials don’t all give up their energy with the same enthusiasm. Some try to hoard it, others share it freely. Air, for instance, has a thermal conductivity of about 0.0244, while stainless steel has a value of ~ 6.6. So the pan handle burns you, but the oven air doesn’t—air does not like to let heat move through it, while steel is perfectly happy to move it from Point A to Point B.
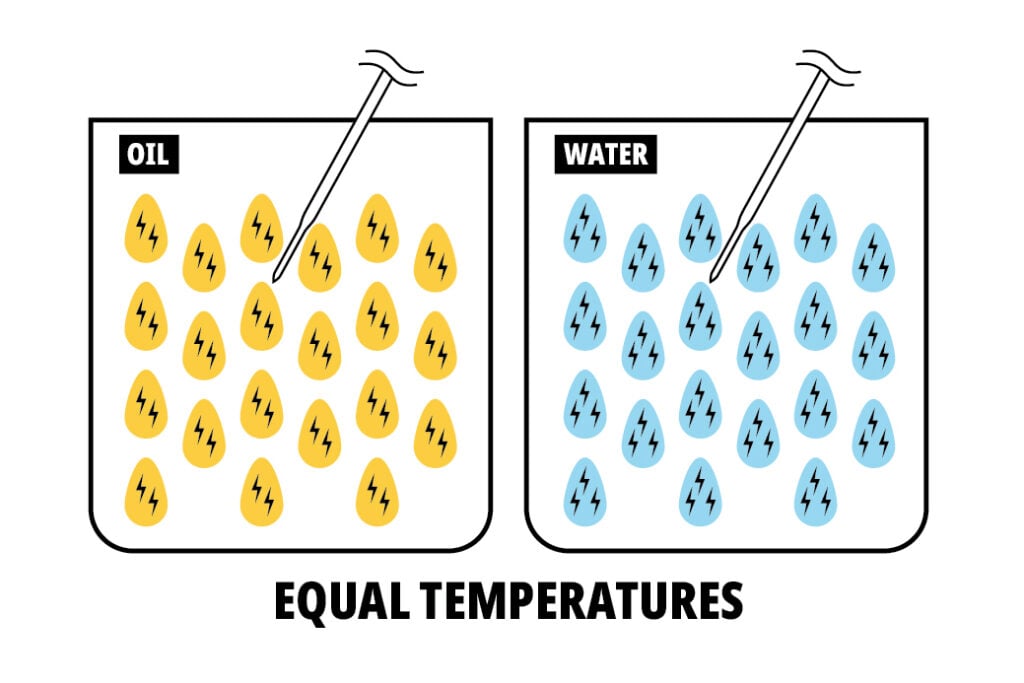
If we compare water and oil we can find another answer to our initial inquiry: Water has a thermal conductivity of ~0.616, but vegetable oil has a conductivity of ~0.17.
So every gram of water contains about 2.5 times as much energy as a gram of oil, and it’s also 3.6 times happier to share that energy with our Thermapen ONE probe. It is a thermodynamic impossibility for oil and water at the same temperature to read at the same speed.
The parable of the new Mercedes
There was once a man of new fortune who was impressed by a commercial while watching golf. So he gave his Honda to his son and bought for himself a new Mercedes. And it happened that he went to a quiet spot on the edge of town and revved the engine and felt joy. But when he floored it, behold, he did not get to 60 in 4.5 seconds. He did not even get to 60 in 6.3. And he returned to the dealership in wrath, but the Dealer said to him “My guy, those numbers are posted by a professional driver on a closed track under specific conditions. Not by you on a gravelly stretch of Highway 15.” And the man drove home in exceeding comfort and luxury, though not on a closed track.
Lab tests set the speed, and it’s still the fastest
Every single Thermapen is individually tested for speed and accuracy in a laboratory setting. If one doesn’t pass the speed test, they get rid of it or try to fix it so that it passes. That lab test is done with running water that is set to a hyper-accurate temperature. This is the thermometry equivalent of a professional driver on a closed track.
The comparison is quite apt. To the same extent that a mid-line commuter car won’t keep up with a sports car on a track, it also won’t keep up on the city roads. And to the extent that Thermapen ONE is the fastest in the lab, it is also the fastest out in the world. And it is the fastest.
Ice bath test for speed
You can, however, take your Thermapen to the racetrack, as it were. If you want to see the speed, you can make an ice bath and test your Thermapen at home.
Fill a cup completely with crushed ice and only fill in the gaps with cold water. Give it a stir, then plunge your Thermapen’s tip into the center of the ice mass. You’ll be shocked at how fast the reading jumps to 32°F (0°C). This is as close to a closed course and professional driver you’re likely to get for this instrument, and the results show its true speed.
What to do with this information…
First, you can use your Thermapen ONE with confidence! Nothing is wrong with it if it takes longer to temp hot oil than water–it’s the fastest, most accurate thermometer you can buy! But beyond that, you can know that when you share this amazing tool with your friends and loved ones, you’re giving them the best tool, one that can change their cooking life forever.
Plus, with a deeper understanding of these thermal principles, we hope you can find more wonder in the world around you!
*All thermal-property values are from information provided on engineertoolbox.com
- It is interesting that air actually has almost twice the specific heat of steel, but because it’s based on energy held per gram, the air in the oven can’t hold nearly as much energy as the pan. There is so little air mass in the oven compared to steel in the handle. Also, air has much lower conductivity. More on that in the next section. ↩︎









